Trending...
- Pacific Harbor Line's Cliatt II Receives Black History Month Trailblazer of the Century Award - 103
- Sellvia Market Enhances Quality Screening for Marketplace Listings
- Transcure Responds to CMS Removal of 285 Inpatient-Only Procedures
Analyst D. Boral Targets NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP) $NRXP at $34 Per Share — Pioneering Breakthroughs in Treatment-Resistant Depression and Chronic Pain
MIAMI - Californer -- NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP), a clinical-stage biopharmaceutical innovator developing breakthrough therapeutics for central nervous system disorders, has emerged as a potential leader in the next generation of psychiatric care. With the global ketamine market projected to expand from $750 million to $3.35 billion by 2034, NRXP is strategically positioned to capture a substantial share through a suite of FDA-designated and investigational treatments aimed at addressing urgent mental health needs.
Revolutionary "ONE-D" Depression Treatment Launches in Florida
NRx recently announced the first-in-Florida launch of its "One Day" (ONE-D) depression treatment, developed in partnership with Ampa Health. The ONE-D protocol uses Ampa's advanced Transcranial Magnetic Stimulation (TMS) device — the first reported technology capable of achieving remission from treatment-resistant depression in a single day.
Unlike conventional 90-day TMS regimens, the ONE-D treatment integrates a single day of precision TMS with D-cycloserine and lisdexamfetamine (both utilized under physician supervision), achieving up to 87% response and 72% remission rates in peer-reviewed nonrandomized studies.
The technology is initially deployed at multiple NRXP HOPE Clinics in Sarasota, Naples, and Fort Myers, with expansion to six Florida locations by year-end 2025 under the direction of Dr. Rebecca Cohen, Medical Director of HOPE Clinics.
More on The Californer
NRX-101: A Breakthrough Therapy for Suicidal Bipolar Depression
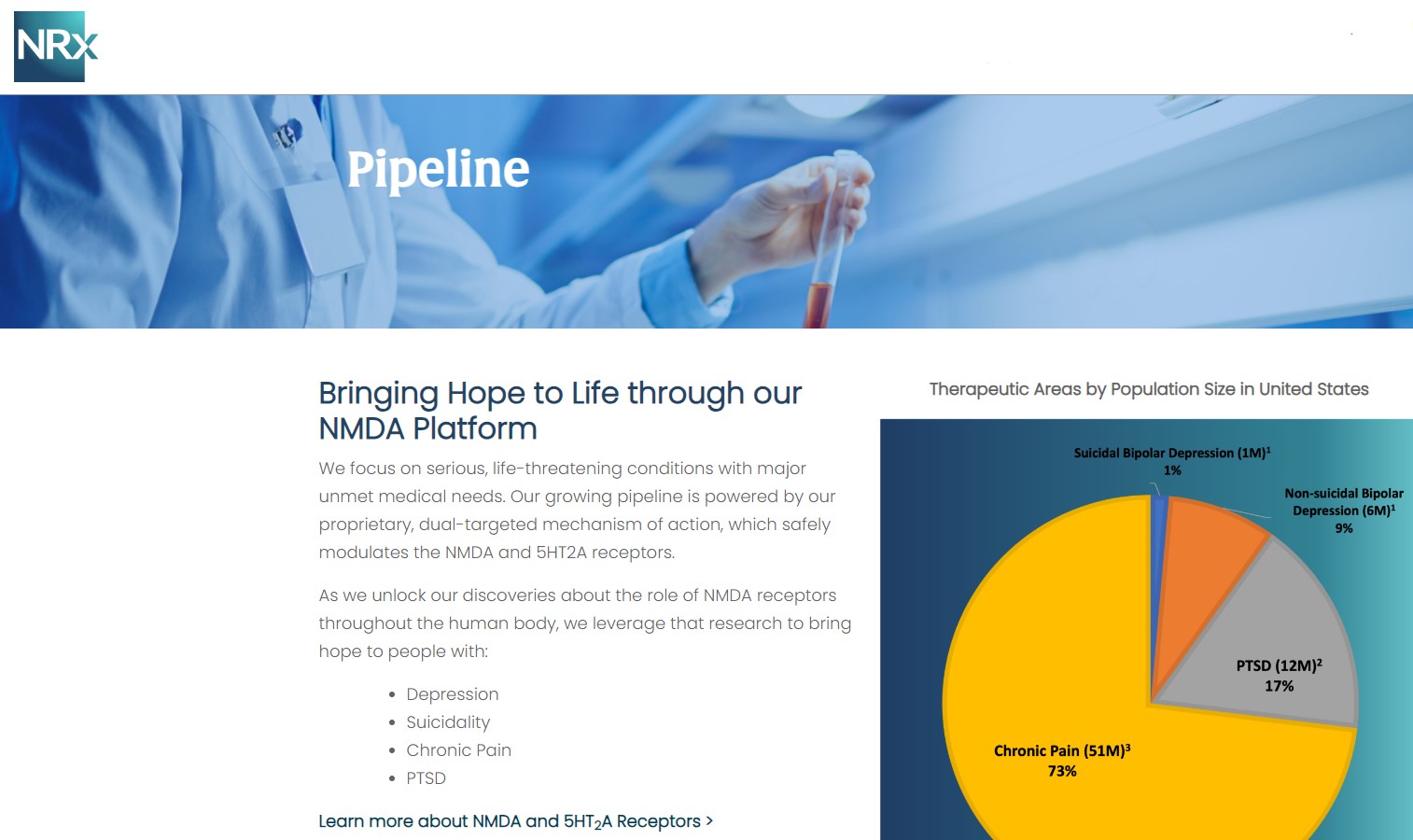
Central to NRx's pipeline is NRX-101, an FDA-designated Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain. The therapy leverages the company's proprietary NMDA platform and is being developed in partnership with Alvogen Pharmaceuticals.
NRX-101 also holds potential as a non-opioid pain treatment and for other indications such as complicated urinary tract infections (UTIs). With over 13 million Americans seriously considering suicide each year (CDC), NRXP's drug candidates could address one of the nation's most pressing health crises.
KETAFREE™: FDA-Focused Push to Modernize Ketamine Treatment
NRXP recently re-filed its Abbreviated New Drug Application (ANDA) for KETAFREE™, a preservative-free IV ketamine formulation, after securing FDA approval of its Suitability Petition. By eliminating benzethonium chloride (BZT) — a preservative linked to neurotoxicity — KETAFREE™ represents a safer, next-generation alternative to traditional ketamine products.
The company has also petitioned the FDA to formally remove BZT from IV formulations, citing expert toxicological data and the growing demand for preservative-free therapies in modern medicine. Given the ongoing ketamine shortage reported by the American Society of Hospital Pharmacists, NRXP's KETAFREE™ could fill a critical supply gap and strengthen its market positioning.
Strategic Acquisitions and Growth Initiatives
NRXP recently completed the acquisition of Dura Medical, an EBITDA-positive network of interventional psychiatry clinics delivering advanced therapies for depression and PTSD. This acquisition, along with pending deals for Neurospa TMS and Cohen & Associates, expands NRXP's reach to more than eight Florida clinic locations, reinforcing its vertically integrated mental health care model.
More on The Californer
To accelerate this rollout, NRXP secured $7.8 million in debt financing from Universal Capital, LLC to fund further HOPE Clinic acquisitions.
Additionally, NRXP has accepted non-binding licensing terms for NRX-100, a drug candidate expected to generate over $300 million in potential milestone payments and tiered double-digit royalties upon commercialization.
Strong Analyst Endorsement
In a recently released report, D. Boral Research issued a Buy rating on NRXP with a $34 price target, citing the company's expanding clinical pipeline, growing footprint in the interventional psychiatry sector, and first-mover advantage in the transformative ONE-D treatment platform.
A Vision for Transforming Mental Health
From its FDA Fast Track designation for IV ketamine (NRX-100) to its breakthrough therapy designation for NRX-101, NRx Pharmaceuticals continues to redefine the boundaries of psychiatric medicine. Its commitment to science-driven innovation — now coupled with real-world clinic deployment through HOPE Clinics — positions NRXP as both a therapeutic pioneer and a potentially lucrative opportunity in the rapidly expanding mental health market.
Company Contact:
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
📍 Wilmington, DE
Matthew Duffy, Chief Business Officer
📧 mduffy@nrxpharma.com | ☎️ (484) 254-6134
🌐 www.nrxpharma.com
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Revolutionary "ONE-D" Depression Treatment Launches in Florida
NRx recently announced the first-in-Florida launch of its "One Day" (ONE-D) depression treatment, developed in partnership with Ampa Health. The ONE-D protocol uses Ampa's advanced Transcranial Magnetic Stimulation (TMS) device — the first reported technology capable of achieving remission from treatment-resistant depression in a single day.
Unlike conventional 90-day TMS regimens, the ONE-D treatment integrates a single day of precision TMS with D-cycloserine and lisdexamfetamine (both utilized under physician supervision), achieving up to 87% response and 72% remission rates in peer-reviewed nonrandomized studies.
The technology is initially deployed at multiple NRXP HOPE Clinics in Sarasota, Naples, and Fort Myers, with expansion to six Florida locations by year-end 2025 under the direction of Dr. Rebecca Cohen, Medical Director of HOPE Clinics.
More on The Californer
- CCHR: While Damaging Antipsychotics Win Approval, Proven Non-Drug Alternatives Remain Ignored
- From Orientation to IEP Meetings: How AI Is Helping Schools Build Stronger Communities
- Arcuri Group Announces Long‑Term Partnership with WakeMed Health & Hospitals to Deliver Situational Awareness and De‑escalation Training
- California: As Trump tears apart decades of environmental progress, Governor Newsom restores nearly 300,000 acres of habitat and cuts average permitting time to 42 days
- New Rock Music Release "Groove Train"
NRX-101: A Breakthrough Therapy for Suicidal Bipolar Depression
Central to NRx's pipeline is NRX-101, an FDA-designated Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain. The therapy leverages the company's proprietary NMDA platform and is being developed in partnership with Alvogen Pharmaceuticals.
NRX-101 also holds potential as a non-opioid pain treatment and for other indications such as complicated urinary tract infections (UTIs). With over 13 million Americans seriously considering suicide each year (CDC), NRXP's drug candidates could address one of the nation's most pressing health crises.
KETAFREE™: FDA-Focused Push to Modernize Ketamine Treatment
NRXP recently re-filed its Abbreviated New Drug Application (ANDA) for KETAFREE™, a preservative-free IV ketamine formulation, after securing FDA approval of its Suitability Petition. By eliminating benzethonium chloride (BZT) — a preservative linked to neurotoxicity — KETAFREE™ represents a safer, next-generation alternative to traditional ketamine products.
The company has also petitioned the FDA to formally remove BZT from IV formulations, citing expert toxicological data and the growing demand for preservative-free therapies in modern medicine. Given the ongoing ketamine shortage reported by the American Society of Hospital Pharmacists, NRXP's KETAFREE™ could fill a critical supply gap and strengthen its market positioning.
Strategic Acquisitions and Growth Initiatives
NRXP recently completed the acquisition of Dura Medical, an EBITDA-positive network of interventional psychiatry clinics delivering advanced therapies for depression and PTSD. This acquisition, along with pending deals for Neurospa TMS and Cohen & Associates, expands NRXP's reach to more than eight Florida clinic locations, reinforcing its vertically integrated mental health care model.
More on The Californer
- JiT Home Buyers Announces Standardized Nationwide Operating Model to Strengthen Homeowner Experience
- Life Hacks of the Rich and Famous named "Best Self Help Podcast"
- Seth Neblett's Mothership Connected: Focuses on P-Funk's Women as Mothership Celebrates 50 Years
- Passive Appoints Ana Bolčević as Head of Design
- At 25, She Became One of the Youngest AAPI Female Founders to Win One of the World's Most Prestigious Design Awards for a Lamp That Makes You Smile
To accelerate this rollout, NRXP secured $7.8 million in debt financing from Universal Capital, LLC to fund further HOPE Clinic acquisitions.
Additionally, NRXP has accepted non-binding licensing terms for NRX-100, a drug candidate expected to generate over $300 million in potential milestone payments and tiered double-digit royalties upon commercialization.
Strong Analyst Endorsement
In a recently released report, D. Boral Research issued a Buy rating on NRXP with a $34 price target, citing the company's expanding clinical pipeline, growing footprint in the interventional psychiatry sector, and first-mover advantage in the transformative ONE-D treatment platform.
A Vision for Transforming Mental Health
From its FDA Fast Track designation for IV ketamine (NRX-100) to its breakthrough therapy designation for NRX-101, NRx Pharmaceuticals continues to redefine the boundaries of psychiatric medicine. Its commitment to science-driven innovation — now coupled with real-world clinic deployment through HOPE Clinics — positions NRXP as both a therapeutic pioneer and a potentially lucrative opportunity in the rapidly expanding mental health market.
Company Contact:
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
📍 Wilmington, DE
Matthew Duffy, Chief Business Officer
📧 mduffy@nrxpharma.com | ☎️ (484) 254-6134
🌐 www.nrxpharma.com
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Source: Corporate Ads
0 Comments
Latest on The Californer
- Long Beach: City to Host Doing Business with the City Expo
- Majestic CA Fire & Disaster Safe Haven/VIP Ranch Retreat w/ Extraordinary Water Resources
- A Gift of Books: Local Business Helps Launch Homeless Library in Lancaster
- Purple Heart Recipient Honored by Hall of Fame Son In Viral Tribute Sparking National Conversation on Service Fatherhood, Healing and Legacy
- DRC Restoration, a Subsidiary of Solid Restoration, Restores Two-Story Home
- iPOP Alum Jacob Batalon Stars in Amazon Prime's "The Wrecking Crew"
- iPOP Alum Olivia Holt Stars in "This Is Not a Test"
- Sellvia Market Enhances Quality Screening for Marketplace Listings
- JiT Home Buyers Strengthens Multi-State Presence as Demand for Flexible Home Selling Solutions Grows
- $3,000,000 Jury Verdict in Police Shooting Case
- Fiz Detailing Launches Professional Car Detailing Services in Fresno, CA
- Transcure Responds to CMS Removal of 285 Inpatient-Only Procedures
- LEVL Launches in over 350 Target Stores Across the East Coast
- Amicly Launches as a Safety-First Social App Designed to Help People Build Real, Meaningful Friendships
- Primeindexer Google indexing platform launched by SEO Danmark APS
- Kaltra Introduces New Downward-Spraying Distribution Technology to Boost Microchannel Evaporator Performance
- Talentica Announces Winners of Multi-Agent Hackathon 2026
- Fritz Coleman's Show "Unassisted Residency" Begins Third Year at El Portal Theatre
- The Good Feet Store Celebrates 25 Years
- Out of the Shadows: JRoc Azael Emerges with Definitive New Album 'Súbele El Volumen"