Trending...
- $5 - $20 Million in Sales for 2026; $25 - $40 Million for 2027 Projected with NASA Agreements; New MOU Signed to Improve Solar Tech in Space - 430
- Only 7 Days Left for Early Bird Registration to the OpenSSL Conference 2025 - 156
- CCHR Warns Global Survey Confirms Electroshock Risks Hidden From Public - 150
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP) $NRXP Has $7.8 Million for Clinic Acquisitions and Purchase of Kadima Neuropsychiatry Institute as Treatment Model and Leading Investigative Site for Suicidal Depression / PTSD
MIAMI - Californer -- Developing NRX-101, an FDA-Designated Investigational Breakthrough Therapy for Suicidal Treatment-Resistant Bipolar Depression and Chronic Pain.
Aiming to be the First FDA-Approved Medication to Treat Suicidal Depression
Designed to Help Address the Needs of Over 13 Million Americans who Seriously Consider Suicide Each Year (CDC).
Applied for New Commissioner's National Priority Vouchers (CNPV) for NRX-100 With Anticipated Decisions on Drug Approval by Year-End.
Application Under CNPV Program is Accretive to Already-Filed Abbreviated New Drug Application With Proprietary Formulation Under Priority Review Request.
Abbreviated New Drug Application Filed for Preservative-Free IV Ketamine.
Ketamine Market Estimated at $750 Million and Projected to Reach $3.35 Billion Globally in 2034.
$7.8 Million Debt Financing to Fuel NRXP HOPE Clinic Acquisitions with
Universal Capital, LLC.
Added to Previously Announced Term Sheet with a Strategic Investor, NRXP HOPE Funding of $10.3 Million is Planned in Near Term.
Agreement to Purchase Kadima Neuropsychiatry Institute Expected to Serve as Clinical Model for Treatment Offerings Nationwide.
Kadima is a Leading Investigative Site for CNS and Psychedelic Research, Having Served as the Lead Site in Nearly All Major Trials in This Space.
Dr. David Feifel, Nationally Recognized Pioneer in Interventional Psychiatry to join as Chief Medical Innovation Officer.
New Drug Application for Treatment of Suicidal Depression; Planned NDA for Accelerated Approval for Bipolar Depression in People at Risk of Akathisia.
Accepted Non-Binding Potential Terms to License and Distribute NRX-100 Drug Providing Over $300 Million in Milestones Plus Tiered Double-Digit Royalties.
Application to Uplist to NASDAQ Global Market from NASDAQ Capital Market.
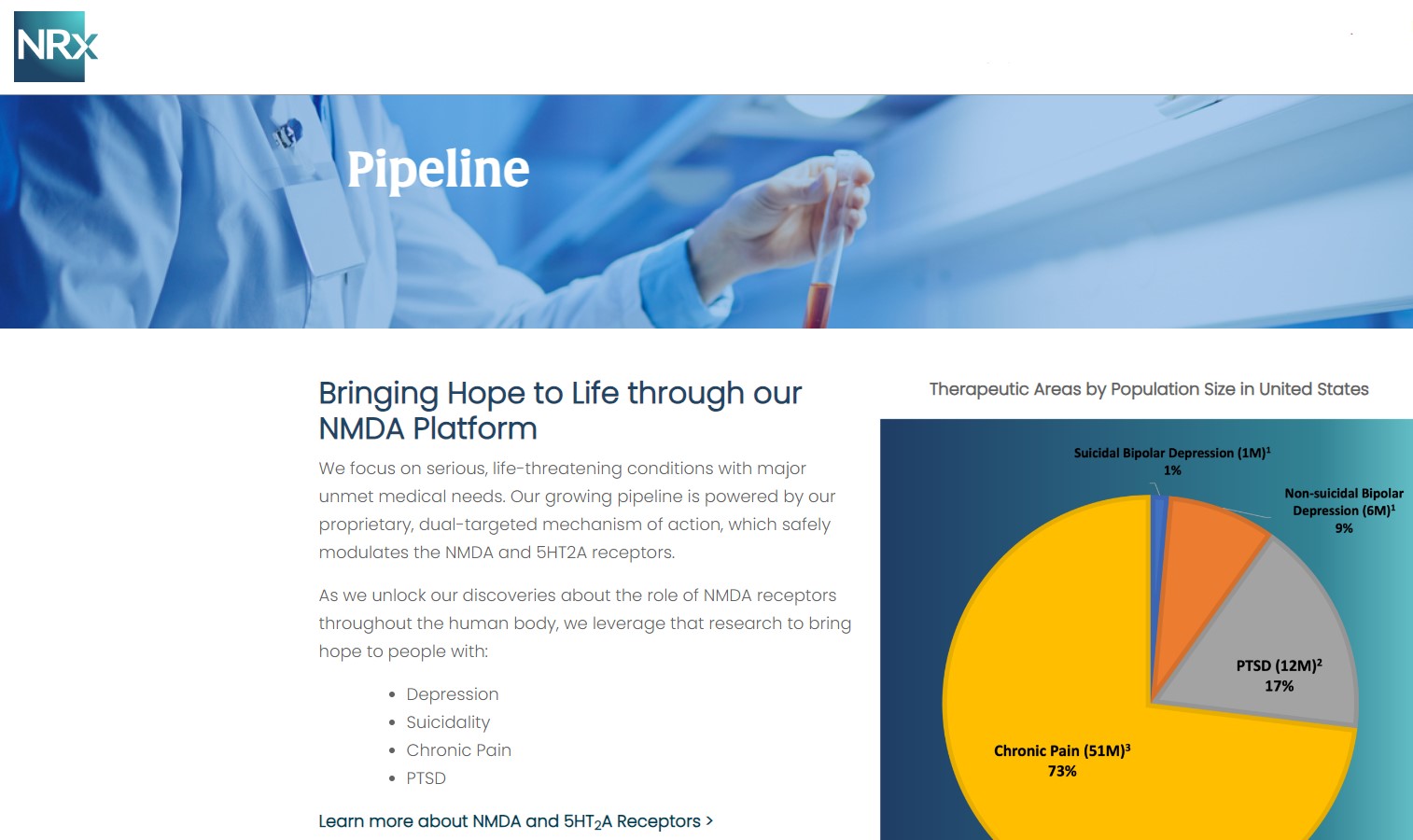
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP) is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. NRXP is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain
NRXP has partnered with Alvogen Pharmaceuticals around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRXP is working on a New Drug Application for NRX-100 (IV ketamine) in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRXP was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
Intravenous ketamine is widely accepted as a standard of care for acute treatment of suicidal depression, in the absence of an FDA-labeled product; the only treatment currently approved by the FDA is electroconvulsive therapy (ECT). According to the CDC, 3.8 million Americans make a plan for suicide each year.
More on The Californer
Filing of Commissioner's National Priority Voucher Application for Intravenous Ketamine (NRX-100)
On June 23rd NRXP announced filing for the newly-announced FDA Commissioner's National Priority Voucher program on behalf of NRX-100, its patent-pending, preservative-free formulation of ketamine for intravenous use.
On June 17, 2025, FDA Commissioner Marty Makary, MD, MPH announced a new approval pathway, the Commissioner's National Priority Voucher (CPNV)1, for approval of drugs to enhance the health interests of Americans. Previously, on May 25 he identified psychedelic drugs for treatment of suicidal depression and PTSD as a national priority.2 The new voucher may be redeemed by drug developers to participate in a Commissioner-led program that shortens its review time from approximately 10-12 months to 1-2 months following a sponsor's final drug application submission.
To qualify for the CNPV, sponsors must submit the chemistry, manufacturing, controls (CMC) portion of the application and the draft labeling at least 60 days before submitting the final application. NRXP has already submitted the CMC portion for NRX-100 and received FDA feedback.
Concurrent with the CNPV process, NRXP is preparing a citizen petition to seek withdrawal of preservative-containing forms of ketamine, based on the toxicity associated with the benzethonium chloride preservative used in the historic formulation. NRXP has also filed a patent on its preservative-free manufacturing process. Approval of either the citizen petition, or the patent, would be expected to enable NRXP to gain market share in the current $750 million generic ketamine market that is forecast to reach $3-5 billion annually by 2033, in addition to a share of the market already established for ketamine products for treating depression.
Strategic Investor Relations Partnership with astr partners
On June 16th NRXP announced a strategic investor relations partnership with astr partners, a boutique investor relations and capital advisory firm focused on the life sciences sector.
astr partners will work with the NRXP and HOPE subsidiary executive teams to execute a comprehensive investor relations program that includes investor targeting, message development, earnings preparation, conference support, and proactive investor engagement.
Abbreviated New Drug Application (ANDA) for Preservative-Free IV Ketamine
On June 5th NRXP announced the transmission of its Abbreviated New Drug Application (ANDA) for electronic filing to the U.S. Food and Drug Administration (FDA) for NRX-100, its preservative-free IV ketamine formulation, for use in all existing approved indications such as anesthesia and pain management.
NRXP anticipates filing a citizen's petition with the FDA to remove benzethonium chloride, a known neurotoxic and cytotoxic substance, from presentations of ketamine intended for intravenous use. NRXP believes that the preservative-free feature of NRX-100 will be deemed of benefit to patients because of the known toxicity of closely related benzalkonium chloride in current drug products. Preservatives were originally added to sterile injectable products in an era when a single vial of medication was used to treat multiple patients, a practice no longer allowed in US hospitals. NRXP has demonstrated that there is no need for such preservatives to maintain stability and sterility in ketamine presentations intended for single-patient use. Should the citizen's petition be granted, all formulations of ketamine sold in the US could face a regulatory requirement to be preservative free.
More on The Californer
$7.8 Million Debt Financing to Fuel NRXP HOPE Clinic Acquisitions
On May 15th NRXP announced signing of a term sheet with Universal Capital, LLC to provide $7.8 million in acquisition capital to initiate subsidiary HOPE's planned national rollup of interventional psychiatry clinics, commencing with previously-announced acquisitions of Dura Medical, Kadima, and NeuroSpa. Together with proceeds of a previously announced strategic investment, this financing is anticipated to provide $10.3 million in acquisition capital.
HOPE's three initial acquisitions represent best-in-class clinics that offer neuroplastic treatments including NRXP ketamine and transcranial magnetic stimulation (TMS) to treat patients with severe depression, PTSD, and related central nervous system conditions. Neuroplastic treatments represent a rapidly emerging class of interventions that cause the growth of new connections (synapses) between brain cells that have been shown in multiple clinical trials to relieve symptoms of depression and suicidality. The FDA has approved TMS devices for a number of indications and has approved a nasal form of ketamine for treatment resistant depression. HOPE's parent company, NRXP, is currently applying to the FDA for approval of intravenous preservative-free ketamine to treat suicidal depression.
Agreement to Purchase Kadima Neuropsychiatry Institute; Foundational Acquisition for the NRXP HOPE Network of Interventional Psychiatry Clinics
On May 13th NRXP announced signing of a definitive agreement to purchase the Kadima Neuropsychiatry Institute. Kadima is expected to serve as the clinical model for treatment offerings in NRXP HOPE-acquired clinics and is expected to continue its role as a leading investigative site for research into neuroplastic therapies including psychedelic medications, transcranial magnetic stimulation (TMS), and hyperbaric therapy.
Kadima is one of California's flagship interventional psychiatry clinics and was among the first to bring ketamine treatment for central nervous system (CNS) disorders out of academic research settings and into clinical practice. The clinic offers a full range of treatments for suicidal depression, PTSD and other CNS disorders. Further, Kadima and David Feifel, MD PhD, Founder and Medical Director of Kadima, have served as leaders in clinical trial work on emerging therapies in CNS for top companies in the industry. Importantly, the clinic is profitable and is forecast to continue growth going forward. Dr. Feifel will join NRXP HOPE as its first Chief Medical Innovation Officer upon closing of the acquisition.
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Media Contact
Company Name: NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Contact Person: Matthew Duffy, Chief Business Officer
Company Website: https://www.nrxpharma.com/
Email: mduffy@nrxpharma.com
Phone: (484) 254-6134
Home Country: United States
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Aiming to be the First FDA-Approved Medication to Treat Suicidal Depression
Designed to Help Address the Needs of Over 13 Million Americans who Seriously Consider Suicide Each Year (CDC).
Applied for New Commissioner's National Priority Vouchers (CNPV) for NRX-100 With Anticipated Decisions on Drug Approval by Year-End.
Application Under CNPV Program is Accretive to Already-Filed Abbreviated New Drug Application With Proprietary Formulation Under Priority Review Request.
Abbreviated New Drug Application Filed for Preservative-Free IV Ketamine.
Ketamine Market Estimated at $750 Million and Projected to Reach $3.35 Billion Globally in 2034.
$7.8 Million Debt Financing to Fuel NRXP HOPE Clinic Acquisitions with
Universal Capital, LLC.
Added to Previously Announced Term Sheet with a Strategic Investor, NRXP HOPE Funding of $10.3 Million is Planned in Near Term.
Agreement to Purchase Kadima Neuropsychiatry Institute Expected to Serve as Clinical Model for Treatment Offerings Nationwide.
Kadima is a Leading Investigative Site for CNS and Psychedelic Research, Having Served as the Lead Site in Nearly All Major Trials in This Space.
Dr. David Feifel, Nationally Recognized Pioneer in Interventional Psychiatry to join as Chief Medical Innovation Officer.
New Drug Application for Treatment of Suicidal Depression; Planned NDA for Accelerated Approval for Bipolar Depression in People at Risk of Akathisia.
Accepted Non-Binding Potential Terms to License and Distribute NRX-100 Drug Providing Over $300 Million in Milestones Plus Tiered Double-Digit Royalties.
Application to Uplist to NASDAQ Global Market from NASDAQ Capital Market.
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP) is a clinical-stage biopharmaceutical company developing therapeutics based on its NMDA platform for the treatment of central nervous system disorders, specifically suicidal bipolar depression, chronic pain and PTSD. NRXP is developing NRX-101, an FDA-designated investigational Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain
NRXP has partnered with Alvogen Pharmaceuticals around the development and marketing of NRX-101 for the treatment of suicidal bipolar depression. NRX-101 additionally has potential to act as a non-opioid treatment for chronic pain, as well as a treatment for complicated UTI.
NRXP is working on a New Drug Application for NRX-100 (IV ketamine) in the treatment of suicidal depression, based on results of well-controlled clinical trials conducted under the auspices of the US National Institutes of Health and newly obtained data from French health authorities, licensed under a data sharing agreement. NRXP was awarded Fast Track Designation for development of ketamine (NRX-100) by the US FDA as part of a protocol to treat patients with acute suicidality.
Intravenous ketamine is widely accepted as a standard of care for acute treatment of suicidal depression, in the absence of an FDA-labeled product; the only treatment currently approved by the FDA is electroconvulsive therapy (ECT). According to the CDC, 3.8 million Americans make a plan for suicide each year.
More on The Californer
- At the SUONO SACRO ACADEMY launch pad in Assisi with the artistic direction of ANDREA CECCOMORI
- Meet a Scientologist Imagines the Future with Artist and Visionary Trish Duggan
- Car Wash Coupon in Downey CA is Not Needed for Car Wash in Downey
- Jamonie Lee Drops New Single "What's It Gonna Be?" — Out Now!
- THE LEAGUE: Where Basketball Meets Culture in the Heart of LA — Played at The Surgeon
Filing of Commissioner's National Priority Voucher Application for Intravenous Ketamine (NRX-100)
On June 23rd NRXP announced filing for the newly-announced FDA Commissioner's National Priority Voucher program on behalf of NRX-100, its patent-pending, preservative-free formulation of ketamine for intravenous use.
On June 17, 2025, FDA Commissioner Marty Makary, MD, MPH announced a new approval pathway, the Commissioner's National Priority Voucher (CPNV)1, for approval of drugs to enhance the health interests of Americans. Previously, on May 25 he identified psychedelic drugs for treatment of suicidal depression and PTSD as a national priority.2 The new voucher may be redeemed by drug developers to participate in a Commissioner-led program that shortens its review time from approximately 10-12 months to 1-2 months following a sponsor's final drug application submission.
To qualify for the CNPV, sponsors must submit the chemistry, manufacturing, controls (CMC) portion of the application and the draft labeling at least 60 days before submitting the final application. NRXP has already submitted the CMC portion for NRX-100 and received FDA feedback.
Concurrent with the CNPV process, NRXP is preparing a citizen petition to seek withdrawal of preservative-containing forms of ketamine, based on the toxicity associated with the benzethonium chloride preservative used in the historic formulation. NRXP has also filed a patent on its preservative-free manufacturing process. Approval of either the citizen petition, or the patent, would be expected to enable NRXP to gain market share in the current $750 million generic ketamine market that is forecast to reach $3-5 billion annually by 2033, in addition to a share of the market already established for ketamine products for treating depression.
Strategic Investor Relations Partnership with astr partners
On June 16th NRXP announced a strategic investor relations partnership with astr partners, a boutique investor relations and capital advisory firm focused on the life sciences sector.
astr partners will work with the NRXP and HOPE subsidiary executive teams to execute a comprehensive investor relations program that includes investor targeting, message development, earnings preparation, conference support, and proactive investor engagement.
Abbreviated New Drug Application (ANDA) for Preservative-Free IV Ketamine
On June 5th NRXP announced the transmission of its Abbreviated New Drug Application (ANDA) for electronic filing to the U.S. Food and Drug Administration (FDA) for NRX-100, its preservative-free IV ketamine formulation, for use in all existing approved indications such as anesthesia and pain management.
NRXP anticipates filing a citizen's petition with the FDA to remove benzethonium chloride, a known neurotoxic and cytotoxic substance, from presentations of ketamine intended for intravenous use. NRXP believes that the preservative-free feature of NRX-100 will be deemed of benefit to patients because of the known toxicity of closely related benzalkonium chloride in current drug products. Preservatives were originally added to sterile injectable products in an era when a single vial of medication was used to treat multiple patients, a practice no longer allowed in US hospitals. NRXP has demonstrated that there is no need for such preservatives to maintain stability and sterility in ketamine presentations intended for single-patient use. Should the citizen's petition be granted, all formulations of ketamine sold in the US could face a regulatory requirement to be preservative free.
More on The Californer
- California: Governor Newsom announces appointments 8.28.25
- California: Governor Newsom issues statement on court decision AGAIN ruling Trump's tariffs as unlawful
- Silva Construction Discusses the Benefits of Garage Conversions for Growing Families
- Governor Newsom signs executive order to bolster California's clean energy progress in face of Trump's wrecking ball
- California: Governor Newsom signs new law to provide fair interest for disaster-affected homeowners
$7.8 Million Debt Financing to Fuel NRXP HOPE Clinic Acquisitions
On May 15th NRXP announced signing of a term sheet with Universal Capital, LLC to provide $7.8 million in acquisition capital to initiate subsidiary HOPE's planned national rollup of interventional psychiatry clinics, commencing with previously-announced acquisitions of Dura Medical, Kadima, and NeuroSpa. Together with proceeds of a previously announced strategic investment, this financing is anticipated to provide $10.3 million in acquisition capital.
HOPE's three initial acquisitions represent best-in-class clinics that offer neuroplastic treatments including NRXP ketamine and transcranial magnetic stimulation (TMS) to treat patients with severe depression, PTSD, and related central nervous system conditions. Neuroplastic treatments represent a rapidly emerging class of interventions that cause the growth of new connections (synapses) between brain cells that have been shown in multiple clinical trials to relieve symptoms of depression and suicidality. The FDA has approved TMS devices for a number of indications and has approved a nasal form of ketamine for treatment resistant depression. HOPE's parent company, NRXP, is currently applying to the FDA for approval of intravenous preservative-free ketamine to treat suicidal depression.
Agreement to Purchase Kadima Neuropsychiatry Institute; Foundational Acquisition for the NRXP HOPE Network of Interventional Psychiatry Clinics
On May 13th NRXP announced signing of a definitive agreement to purchase the Kadima Neuropsychiatry Institute. Kadima is expected to serve as the clinical model for treatment offerings in NRXP HOPE-acquired clinics and is expected to continue its role as a leading investigative site for research into neuroplastic therapies including psychedelic medications, transcranial magnetic stimulation (TMS), and hyperbaric therapy.
Kadima is one of California's flagship interventional psychiatry clinics and was among the first to bring ketamine treatment for central nervous system (CNS) disorders out of academic research settings and into clinical practice. The clinic offers a full range of treatments for suicidal depression, PTSD and other CNS disorders. Further, Kadima and David Feifel, MD PhD, Founder and Medical Director of Kadima, have served as leaders in clinical trial work on emerging therapies in CNS for top companies in the industry. Importantly, the clinic is profitable and is forecast to continue growth going forward. Dr. Feifel will join NRXP HOPE as its first Chief Medical Innovation Officer upon closing of the acquisition.
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Media Contact
Company Name: NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Contact Person: Matthew Duffy, Chief Business Officer
Company Website: https://www.nrxpharma.com/
Email: mduffy@nrxpharma.com
Phone: (484) 254-6134
Home Country: United States
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Source: Corporate Ads
0 Comments
Latest on The Californer
- The Best Cartier Love Bracelet Dupes: Luxury Style Without the Price Tag
- AureaVault Launches U.S.-Licensed Cryptocurrency Exchange with Enhanced Security Features
- IOTAP Named to 2025 Inc. 5000 List of America's Fastest-Growing Private Companies
- California: Governor Newsom issues legislative update 8.28.25
- TEAMSTERS CALIFORNIA KICKS OFF WAVE OF ACTION ON WORKERS' PRIORITIES: GOOD JOBS, AFFORDABILITY, AI ACCOUNTABILITY
- Lineus Medical and Venture Medical Sign New Zealand Distribution Agreement
- Calling All Music Lovers! Get ready to Celebrate One-Hit Wonder Day with Big Bucks & Run For Office
- Black Plumbing Expands to Cleburne, TX, Bringing Over 30 Years of Trusted Plumbing Service
- Rising Talent Mckenna Bridger with Lead Role in Upcoming Film Nightmare
- Emerging Talent Mckenna Bridger on the Rise with Lead in Upcoming Film Nightmare
- TWO ERVIN COHEN & JESSUP PARTNERS NAMED "LEADING REAL ESTATE LAWYERS" BY LAWDRAGON
- City of Long Beach Launches Community Survey for the Urban Forest Plan
- Governor Newsom deploys new teams to fight crime in major California cities, building on existing successful efforts
- From Horror to Heartfelt: How Author Cassondra Windwalker Shifts Gears with her Latest Novel
- $5 - $20 Million in Sales for 2026; $25 - $40 Million for 2027 Projected with NASA Agreements; New MOU Signed to Improve Solar Tech in Space
- Healthcare Inflation Ahead: Strategies to Strengthen Your Boeing Retirement
- ERC Marketplace Graduates to Debut New Products at Menifee Foodie Friday September 5
- New Book: Cold War Sci-Fi Thriller Arrives Today
- BeeCool Bikes Unveils Next-Generation "Super Frame" with Bee Defender Series
- Elklook Redefines Eyewear: Beyond Fashion, Creating Value