Trending...
- Pacific Harbor Line's Cliatt II Receives Black History Month Trailblazer of the Century Award - 134
- Transcure Responds to CMS Removal of 285 Inpatient-Only Procedures - 121
- JiT Home Buyers Strengthens Multi-State Presence as Demand for Flexible Home Selling Solutions Grows - 115
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP) $NRXP has Entered into a Joint Offering with neurocare Group for Neuroplastic Therapy Targeting Depression, PTSD and Other Mental Health Afflictions
MIAMI - Californer -- As the mental health crisis deepens in the United States, few biotech companies are as directly aligned with both urgent unmet medical need and near-term regulatory catalysts as NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP). With groundbreaking real-world data from over 70,000 patients, multiple FDA pathways underway, a newly debt-free balance sheet, and an expanding neuroplastic therapy ecosystem, NRXP is positioning itself as a potentially transformative force in the treatment of suicidal depression, bipolar depression, PTSD, and chronic pain.
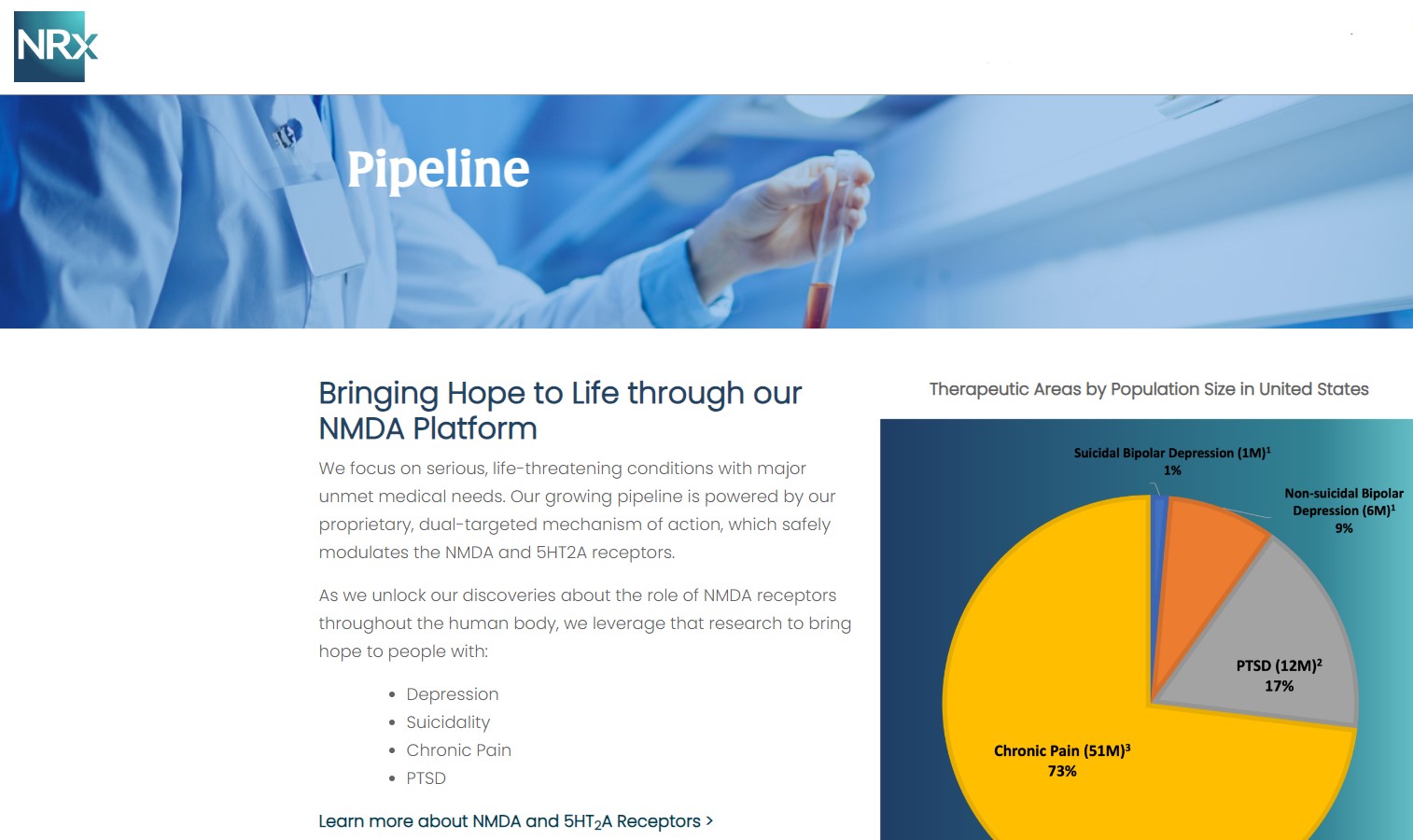
A Massive, Unmet Need with No Approved Drug Solution
According to the CDC, more than 13 million Americans seriously consider suicide each year, yet no medication is currently FDA-approved to treat suicidal ideation. Today, electroconvulsive therapy (ECT) remains the only approved intervention—an invasive option often reserved as a last resort.
NRXP is attempting to change that paradigm.
The company is advancing NRX-100, a preservative-free intravenous ketamine, under FDA Fast Track designation for the treatment of suicidal depression and bipolar depression. Importantly, NRXP recently licensed Real World Evidence (RWE) data from over 70,000 U.S. patients, marking one of the largest datasets ever assembled for ketamine use in suicidality.
70,000-Patient Ketamine Dataset Headed to the FDA
On January 14, NRXP announced plans to submit this expansive real-world dataset to the FDA in support of Accelerated Approval of NRX-100.
Preliminary analysis of a 20,000-patient subset revealed:
The full 70,000-patient analysis will be presented to regulators, strengthening NRXP's case that ketamine—when delivered in a safer, preservative-free formulation—may finally offer a pharmacologic option for acute suicidality.
More on The Californer
If successful, NRXP could help bring the first FDA-approved drug for suicidal ideation to market.
KETAFREE™: A Cleaner Ketamine with a Clear Regulatory Path
Parallel to NRX-100, NRXP is pursuing approval of KETAFREE™, a preservative-free IV ketamine via an Abbreviated New Drug Application (ANDA). In December, the FDA confirmed the ANDA is "substantially complete" and assigned a PDUFA goal date of July 29, 2026.
Why this matters:
Approval of KETAFREE™ could establish NRXP as a differentiated supplier in a large, existing market—separate from the novel drug opportunity represented by NRX-100.
NRX-101: A Breakthrough Therapy with Expanding Potential
NRXP's flagship pipeline asset, NRX-101, is an FDA-designated Breakthrough Therapy for suicidal treatment-resistant bipolar depression. The oral combination drug (D-cycloserine + lurasidone) was specifically engineered to deliver neuroplastic benefits while mitigating hallucination risk—an innovation protected by composition-of-matter patents worldwide.
In late 2024, NRXP expanded NRX-101's potential by adding a new pipeline indication: augmentation of Transcranial Magnetic Stimulation (TMS).
Recent real-world and clinical data suggest:
With projections that over 1 million Americans per year may receive TMS by 2030, this new indication opens a previously unanticipated commercial pathway for NRX-101.
Building a Neuroplastic Therapy Ecosystem with neurocare
In January, NRXP announced a joint initiative with neurocare Group AG to create a nationwide network of integrated neuroplastic therapy clinics targeting depression, PTSD, and other serious mental health disorders.
More on The Californer
The model combines:
The rollout will leverage:
Early pilot programs—particularly among first responders with PTSD and depression—have demonstrated exceptionally high remission rates, positioning NRXP at the intersection of drug development and scalable clinical delivery.
A Clean Balance Sheet and Analyst Validation
In December, NRXP eliminated 100% of its balance-sheet debt, converting $5.4 million into equity with no additional warrants—a notable reset that strengthens the company's financial footing ahead of key regulatory milestones.
Adding to investor confidence, D. Boral Capital issued a Buy rating with a $34 price target, citing NRXP's pipeline breadth, regulatory momentum, and differentiated mental health strategy.
The Bottom Line
NRx Pharmaceuticals stands out in a crowded biotech landscape by targeting:
As regulatory submissions advance and clinical integration expands, NRXP may be approaching an inflection point—one with the potential to redefine how suicidal depression and treatment-resistant mental illness are treated in the U.S.
Ticker: N A S D A Q: NRXP
More Information:
For more information:
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Website: www.nrxpharma.com
Media Contact: Matthew Duffy, Chief Business Officer
Email: mduffy@nrxpharma.com | Phone: (484) 254-6134
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
A Massive, Unmet Need with No Approved Drug Solution
According to the CDC, more than 13 million Americans seriously consider suicide each year, yet no medication is currently FDA-approved to treat suicidal ideation. Today, electroconvulsive therapy (ECT) remains the only approved intervention—an invasive option often reserved as a last resort.
NRXP is attempting to change that paradigm.
The company is advancing NRX-100, a preservative-free intravenous ketamine, under FDA Fast Track designation for the treatment of suicidal depression and bipolar depression. Importantly, NRXP recently licensed Real World Evidence (RWE) data from over 70,000 U.S. patients, marking one of the largest datasets ever assembled for ketamine use in suicidality.
70,000-Patient Ketamine Dataset Headed to the FDA
On January 14, NRXP announced plans to submit this expansive real-world dataset to the FDA in support of Accelerated Approval of NRX-100.
Preliminary analysis of a 20,000-patient subset revealed:
- Rapid resolution of depression and suicidality
- Clinical responses consistent with prior randomized NIH-sponsored trials
- Outcomes that compare favorably to currently approved antidepressant products
The full 70,000-patient analysis will be presented to regulators, strengthening NRXP's case that ketamine—when delivered in a safer, preservative-free formulation—may finally offer a pharmacologic option for acute suicidality.
More on The Californer
- Governor Newsom, First Partner Siebel Newsom announce new inductees to the California Hall of Fame
- Spring Surge in 55+ Communities: What Buyers and Sellers Need to Know in 2026
- Jason Caras Launches The Caras Institute Following Successful Exit from IT Authorities
- NuCicer & Stricks Ag Launch Strategic Partnership to Scale High-Protein Chickpeas to Meet Immediate
- Serina Damesworth Hired as Century Fasteners Corp. – Director of Quality
If successful, NRXP could help bring the first FDA-approved drug for suicidal ideation to market.
KETAFREE™: A Cleaner Ketamine with a Clear Regulatory Path
Parallel to NRX-100, NRXP is pursuing approval of KETAFREE™, a preservative-free IV ketamine via an Abbreviated New Drug Application (ANDA). In December, the FDA confirmed the ANDA is "substantially complete" and assigned a PDUFA goal date of July 29, 2026.
Why this matters:
- Current ketamine products contain benzethonium chloride (BZT), a preservative not recognized as safe by the FDA
- KETAFREE™ eliminates this additive
- The global ketamine market is estimated at $750 million annually
- Manufactured in the U.S., aligning with MAHA initiatives to remove toxic substances from medicines and strengthen domestic supply chains
Approval of KETAFREE™ could establish NRXP as a differentiated supplier in a large, existing market—separate from the novel drug opportunity represented by NRX-100.
NRX-101: A Breakthrough Therapy with Expanding Potential
NRXP's flagship pipeline asset, NRX-101, is an FDA-designated Breakthrough Therapy for suicidal treatment-resistant bipolar depression. The oral combination drug (D-cycloserine + lurasidone) was specifically engineered to deliver neuroplastic benefits while mitigating hallucination risk—an innovation protected by composition-of-matter patents worldwide.
In late 2024, NRXP expanded NRX-101's potential by adding a new pipeline indication: augmentation of Transcranial Magnetic Stimulation (TMS).
Recent real-world and clinical data suggest:
- 87% clinical response
- 72% remission
- Achieved after a single day of TMS combined with oral D-cycloserine
With projections that over 1 million Americans per year may receive TMS by 2030, this new indication opens a previously unanticipated commercial pathway for NRX-101.
Building a Neuroplastic Therapy Ecosystem with neurocare
In January, NRXP announced a joint initiative with neurocare Group AG to create a nationwide network of integrated neuroplastic therapy clinics targeting depression, PTSD, and other serious mental health disorders.
More on The Californer
- JiT Home Buyers Emphasizes Transparency and Clear Communication in Direct Home Sales Nationwide
- California: Governor Newsom announces 95% compliance with prohibition of illegal kratom products
- ReProp Financial Announces the Hiring of Veteran Commercial Lender Mark Hunton
- National Expansion Ignited Across Amazon $AMZN, Chewy $CHWY & Walmart $WMT: NDT Pharmaceuticals, Inc. (Stock Symbol: NDTP) $NDTP
- Breathe Biologics, Inc. names Ed Johnson as Chief Executive Officer
The model combines:
- TMS
- Ketamine and other neuroplastic drugs
- Hyperbaric oxygen therapy
- Psychotherapy
The rollout will leverage:
- neurocare's existing clinic footprint
- HOPE Therapeutics clinics
- 400+ Apollo® TMS machines already deployed nationwide
Early pilot programs—particularly among first responders with PTSD and depression—have demonstrated exceptionally high remission rates, positioning NRXP at the intersection of drug development and scalable clinical delivery.
A Clean Balance Sheet and Analyst Validation
In December, NRXP eliminated 100% of its balance-sheet debt, converting $5.4 million into equity with no additional warrants—a notable reset that strengthens the company's financial footing ahead of key regulatory milestones.
Adding to investor confidence, D. Boral Capital issued a Buy rating with a $34 price target, citing NRXP's pipeline breadth, regulatory momentum, and differentiated mental health strategy.
The Bottom Line
NRx Pharmaceuticals stands out in a crowded biotech landscape by targeting:
- One of the most urgent unmet needs in medicine
- With unprecedented real-world clinical data
- Multiple FDA pathways (Fast Track, ANDA, Breakthrough Therapy)
- A growing neuroplastic therapy infrastructure
- And a now debt-free balance sheet
As regulatory submissions advance and clinical integration expands, NRXP may be approaching an inflection point—one with the potential to redefine how suicidal depression and treatment-resistant mental illness are treated in the U.S.
Ticker: N A S D A Q: NRXP
More Information:
For more information:
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Website: www.nrxpharma.com
Media Contact: Matthew Duffy, Chief Business Officer
Email: mduffy@nrxpharma.com | Phone: (484) 254-6134
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Source: CorporateAds
0 Comments
Latest on The Californer
- Progressive Dental & The Closing Institute Partner with Zest Dental Solutions to Elevate Full-Arch Growth and Patient Outcomes
- CCHR: While Damaging Antipsychotics Win Approval, Proven Non-Drug Alternatives Remain Ignored
- From Orientation to IEP Meetings: How AI Is Helping Schools Build Stronger Communities
- Arcuri Group Announces Long‑Term Partnership with WakeMed Health & Hospitals to Deliver Situational Awareness and De‑escalation Training
- California: As Trump tears apart decades of environmental progress, Governor Newsom restores nearly 300,000 acres of habitat and cuts average permitting time to 42 days
- New Rock Music Release "Groove Train"
- JiT Home Buyers Announces Standardized Nationwide Operating Model to Strengthen Homeowner Experience
- Life Hacks of the Rich and Famous named "Best Self Help Podcast"
- Seth Neblett's Mothership Connected: Focuses on P-Funk's Women as Mothership Celebrates 50 Years
- Passive Appoints Ana Bolčević as Head of Design
- At 25, She Became One of the Youngest AAPI Female Founders to Win One of the World's Most Prestigious Design Awards for a Lamp That Makes You Smile
- Sunnyvale Family and Cosmetic Dentistry Highlights Community Access to Advanced Laser Dentistry
- Juego Studios Extends Full-Cycle Game Development & Outsourcing Capabilities to the UAE Market
- Composer Aidan Vass Releases Cleome, a Generative Ambient Music Engine for Spas and Wellness Centers
- Enhanced Perimeter Protection Now Available in Ashville Through Countrywide Rental
- iPOP Alum Elizabeth Zacero competing in Survivor Croatia
- Mento Labs LLC Launches ForgeFit, an AI-Powered Fitness Coaching App for Everyone
- How to Add Phone Calls to OpenClaw Easily with Call24x7
- VENUS Goes Live on CATEX Exchange As UK Financial Ltd Activates The Premier Division Of The Maya Meme's League
- EDCAPIT Raises Investment at $20M Valuation Cap; Khayrullaev Invests and Joins as Advisor