Trending...
- California: Governor Newsom, Superintendent Thurmond announce over $618 million to support another 458 community schools
- $4.3 Million Patent Application Waiver Fee Granted by FDA on New Drug Application Fee for Treatment Addressing Suicidal Depression & PTSD: NRX Pharma
- xREnergy up as much as +3,094,634% on first day listed on the XRP Ledger. Ticker : $XRE
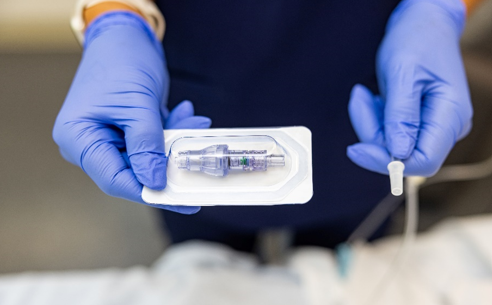
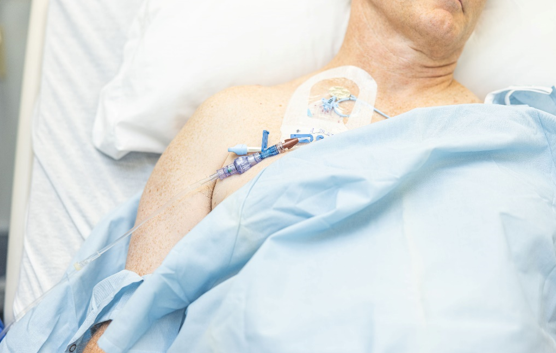
FAYETTEVILLE, Ark. - Californer -- Lineus Medical® is proud to announce the U.S. Food and Drug Administration (FDA) has cleared its flag ship product, SafeBreak® Vascular, for use on all vascular access lines including peripheral IV catheters, midlines, peripherally inserted central catheters, central venous catheters, IV ports, and interosseous vascular access devices on patients two weeks of age and up. This allows SafeBreak Vascular to be installed on both adults and children and on every type of IV line.
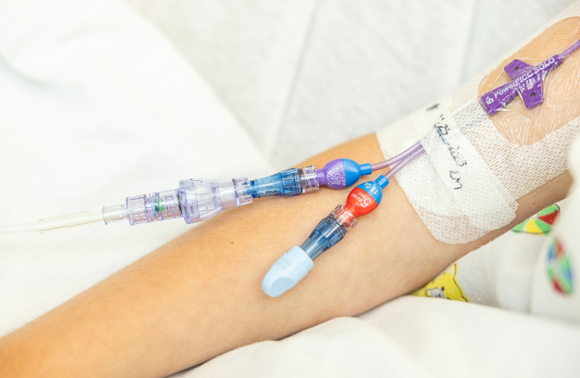
SafeBreak Vascular is the only breakaway device for IV lines proven to reduce IV complications.1 When a harmful force is placed on the line, SafeBreak intentionally separates to remove the damaging force and protect the patient's IV. When SafeBreak separates, valves on both ends of the device close preventing medicine spills from the pump and blood loss from the patient. The separated device also causes the IV pump to alarm, notifying the nursing staff to check on the patient. To replace SafeBreak, each component is unthreaded from the line, a new sterile SafeBreak is installed, and the patient's infusion is restarted. Patients avoid additional needlesticks, nurses save time, and hospitals save money1. It's a win-win-win.
More on The Californer
Ryan Brick, VP of Sales, stated, "This is a large milestone for our company and a steppingstone towards our mission of reducing IV-line failure. With SafeBreak's clearance for all vascular lines, we can help protect every patient user and every IV line throughout the entire country. Sales momentum has continued to build since our pediatric clearance this past May, and now, the expansion of SafeBreak's indications will bring even more interest and help introduce the product to more hospitals. We will continue to work to make SafeBreak the standard of care for every hospital and patient."
Vance Clement, CEO of Lineus Medical, stated, "What an exciting time for SafeBreak Vascular and Lineus Medical! We recently gained licence in Canada and now FDA clearance for all medical lines for patients to two weeks of age and up in the United States. SafeBreak is a simple device that can make a big difference for nurses and hospitals. These new indications are for more complicated and more expensive IV lines that are used for long-term infusions, cancer patients, and in emergency situations. SafeBreak has been shown to work well on peripheral IVs, and now it can be used by nurses to protect these more expensive lines on more vulnerable patient populations. With these additional indications, we can expand the benefit of SafeBreak throughout the entire hospital."
More on The Californer
ABOUT LINEUS MEDICAL
Lineus Medical is the developer of a break-away technology that is proven to reduce IV restarts and IV complications1. Our mission is to remove the pains associated with medical lines. More information about Lineus Medical can be found on the company website: www.lineusmed.com or you can follow the company on Facebook or LinkedIn.
1. Data on File
MKG-0237 Rev 00
SafeBreak Vascular is the only breakaway device for IV lines proven to reduce IV complications.1 When a harmful force is placed on the line, SafeBreak intentionally separates to remove the damaging force and protect the patient's IV. When SafeBreak separates, valves on both ends of the device close preventing medicine spills from the pump and blood loss from the patient. The separated device also causes the IV pump to alarm, notifying the nursing staff to check on the patient. To replace SafeBreak, each component is unthreaded from the line, a new sterile SafeBreak is installed, and the patient's infusion is restarted. Patients avoid additional needlesticks, nurses save time, and hospitals save money1. It's a win-win-win.
More on The Californer
- Poll Finds Overwhelming Opposition to Keeping Big Cats as Pets
- SM Telecom Expands AT&T Partnership to Deliver Cutting-Edge 5G+ Wireless Solutions New Collab Brings AT&T's Advanced 5G+ Technology to Cellphone
- Governor Newsom proclaims Older Californians Month
- Groundbreaking Launches 154-Acre Los Cerritos Wetlands Restoration in Long Beach – Single Largest Increase in Open Space in Long Beach in Decades
- Don Barnhart Drops Unapologetically Hilarious Comedy Special "You Do You," On Open Bar Network
Ryan Brick, VP of Sales, stated, "This is a large milestone for our company and a steppingstone towards our mission of reducing IV-line failure. With SafeBreak's clearance for all vascular lines, we can help protect every patient user and every IV line throughout the entire country. Sales momentum has continued to build since our pediatric clearance this past May, and now, the expansion of SafeBreak's indications will bring even more interest and help introduce the product to more hospitals. We will continue to work to make SafeBreak the standard of care for every hospital and patient."
Vance Clement, CEO of Lineus Medical, stated, "What an exciting time for SafeBreak Vascular and Lineus Medical! We recently gained licence in Canada and now FDA clearance for all medical lines for patients to two weeks of age and up in the United States. SafeBreak is a simple device that can make a big difference for nurses and hospitals. These new indications are for more complicated and more expensive IV lines that are used for long-term infusions, cancer patients, and in emergency situations. SafeBreak has been shown to work well on peripheral IVs, and now it can be used by nurses to protect these more expensive lines on more vulnerable patient populations. With these additional indications, we can expand the benefit of SafeBreak throughout the entire hospital."
More on The Californer
- Cygnet Theatre Announces The Cast and Creative Team of Rodgers and Hammerstein's Oklahoma!
- Shon Garage Door Repair Expands Trusted Services to San Diego, CA
- Nicky Dare Honors Indonesian Heritage at VAPI–Valley Asian and Pacific Islanders Cultural Festival
- California launches new AI-powered chatbot that provides wildfire resources in 70 languages
- Summit Startup Solutions Launches Innovative, Flexible Support Services for Startups
ABOUT LINEUS MEDICAL
Lineus Medical is the developer of a break-away technology that is proven to reduce IV restarts and IV complications1. Our mission is to remove the pains associated with medical lines. More information about Lineus Medical can be found on the company website: www.lineusmed.com or you can follow the company on Facebook or LinkedIn.
1. Data on File
MKG-0237 Rev 00
Source: Lineus Medical
0 Comments
Latest on The Californer
- California: Governor Newsom issues statement on Pope Leo XIV, the first American Pope
- New poll shows high rates of employee burnout amid concerns over politics and personal finances
- Tessellations Appoints Luthern Williams as Head of School
- Aureli Construction Sets the Standard for Seamless Home Additions in Greater Boston
- Psychological Thriller "Killing Off Connor" To Open 34th IFS Film Fest After 12-years In Post
- Harvest Properties Acquires Two San Francisco Bay Area Self Storage Facilities for $44.2 Million
- California businesses in near-universal compliance with prohibition of intoxicating hemp products harmful to youth
- California: Governor Newsom announces upgrades to 21 state fish hatcheries to boost salmon populations
- Solaris Energy Infrastructure, Inc. (SEI) Investors Who Lost Money Have Opportunity to Lead Securities Fraud Lawsuit
- Risk Rater, Threat Assessment App, gives Users the Same Threat Evaluation as the Rich and Powerful
- Is it Really True That Tariffs Will Raise Car Insurance Rates?
- ScreenPoints Puts Film Investors in the Credits—and in the Money With New FinTech Platform
- Coastal Business Systems Wraps Up Successful 2025 Tech Show in Redding
- AdOcto Turns AirBnBs Into High-Impact Advertising Channels
- Zefr Announces Launch of Pre-Screen Brand Safety Solution for Google's Search Partner Network (SPN)
- Pathways to Adulthood Conference May 17 at Melville Marriott Honoring NYS Assembly Member Jodi Giglio, Suffolk County Legislator Nick Caracappa
- Adster Techologies awarded US Patent for breakthrough innovation in reducing latency in Ad Serving
- Flexi-View Lending Closes $5.05 Million Residential Acquisition Loan in Billings, Montana
- Robert Fabbio Inducted into the Austin Technology Council Hall of Fame
- California: Governor Newsom, Superintendent Thurmond announce over $618 million to support another 458 community schools